
This parameter uses the D/H ratio in a planetary sample compared to standard mean ocean water (SMOW): δD = x 1000. The D/H is expressed by a derived parameter called delta D, δD. The value of the D/H ratio as a tracer of potential water sources is shown in the diagram below. The model also considers dissolution of hydrogen into the metallic core of Earth and its effect on D/H. It would not take much dissolution of hydrogen from this atmosphere to modify the hydrogen isotopic composition of the originally accreted Earth to account for the overall hydrogen isotopic composition (given by D/H) of the planet. This primitive atmosphere would have formed by accretion of solar nebula gas, which has a small D/H ratio, about 20 parts per million. (See P S R D article: The Complicated Origin of Earth's Water).) Through detailed modeling they show that a minor but important component came via "ingassing" from a proto-atmosphere surrounding Earth. Jun Wu and colleagues at Arizona State University have investigated potential sources of Earth's water. Estimates of D/H in the bulk Earth and Moon are about 150 parts per million. D/H in the water that is bound inside hydrous minerals, averages 140 parts per million. As a reference, the measured D/H in Earth's standard mean ocean water ( SMOW) is 156 x 10 -6 (0.000156, or 156 parts per million). Its nucleus contains a proton and a neutron hydrogen's nucleus contains only a proton. (See P S R D CosmoSparks report: Water, Carbonaceous Chondrites, and Earth.) The crucial data are the deuterium/ hydrogen ratios (D/H). Where did this water come from? Based on the similarity in hydrogen isotopic composition in Earth and carbonaceous chondrite meteorites, a promising idea is that most water was delivered by impacting planetesimals similar to carbonaceous chondrites. The interior contains about seven oceans' worth, tied up in rocks in the mantle and in the metallic core. The oceans contain 1.4 x 10 21 kilograms of water, but far from all the water in the Earth. Water in the Earth's oceans absorbs the orange and red of the visible electromagnetic spectrum, reflecting the blue, causing the Earth to be a pale blue dot when viewed from space. Water Water Everywhere, But Where Did it Come From? Hydrogen Isotopes in Small Lunar Samples Provide Clues to the Origin of the Earth and Moon - Short Slide Summary (with accompanying notes). (2019) A Unified Model for Hydrogen in the Earth and Moon: No One Expects the Theia Contribution, Geochemistry, v. This scenario has implications for the nature of the giant impact that formed the Moon. This acquisition of solar nebula hydrogen could have taken place in planetary bodies large enough (about 40% of the final mass of the Earth) to form an atmosphere of nebula gas, some of which dissolved into the molten body. Desch and Robinson suggest that the low D/H in the QMDs indicate that low D/H regions exist inside the Moon, perhaps reflecting batches of lunar raw materials that had acquired hydrogen from the inner solar nebula (the disk of gas and dust surrounding the infant Sun), which had D/H of about 20 parts per million. Slowly-cooled igneous rocks (called quartz monzodiorites, QMDs for short) from the Apollo 15 landing site on the Moon have D/H of 40 parts per million, much lower than Earth and carbonaceous chondrites (about 140 parts per million). Most of the hydrogen in both bodies has a deuterium/hydrogen ratio of about 150 parts per million (not much deuterium). 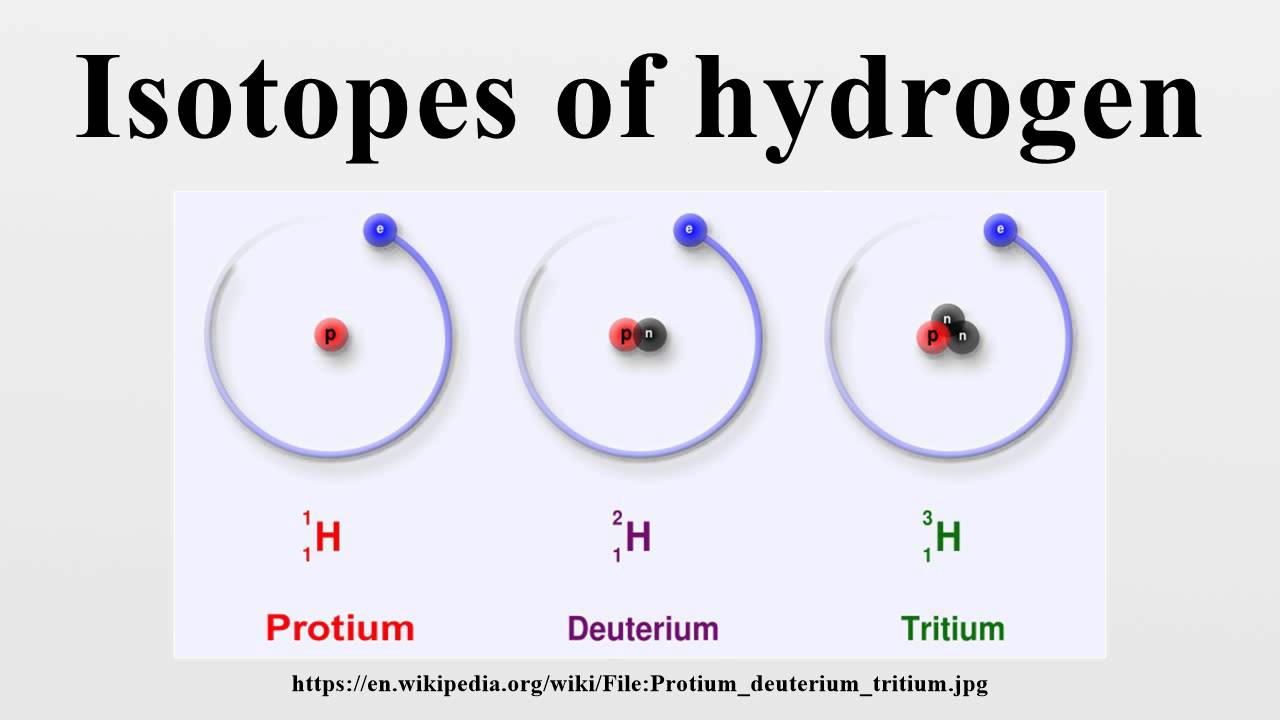
Steven Desch (Arizona State University) and Katharine Robinson (Lunar and Planetary Institute, TX) used the abundances of hydrogen isotopes (hydrogen and deuterium) to probe the sources of water in the Moon and Earth. The difference is almost certainly due to how the two bodies formed. Hawai'i Institute of Geophysics and PlanetologyĪ prominent difference between Earth and the Moon is that one of them has lots of water, making it look like a pale blue dot when viewed from space, and the other is a gray, dusty place. Small samples of igneous rocks from the Moon have low deuterium/hydrogen ratios, indicating that some planetary bodies involved in the formation of the Earth and Moon trapped gas from the solar nebula. Hydrogen Isotopes in Small Lunar Samples Provide Clues to the Origin of the Earth and Moon (March, 2020) Hydrogen Isotopes in Small Lunar Samples Provide Clues to the Origin of the Earth and Moon, PSRD.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
